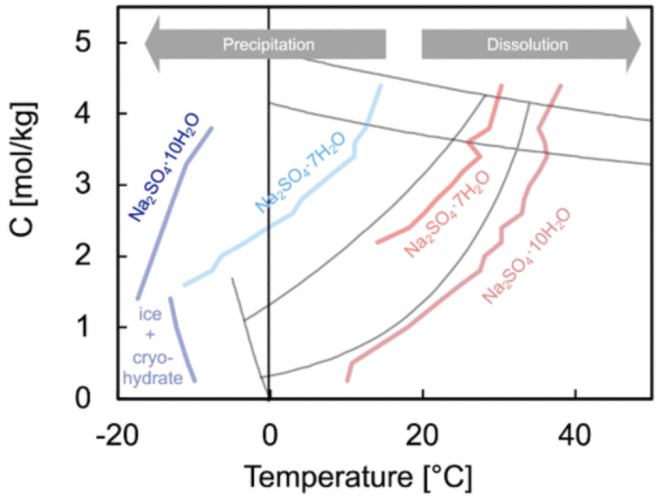
Check out our most recent publication in the Journal of Physical Chemistry C! Sodium sulfate is a promising material platform for thermal energy storage, where the giant latent heat of a phase transition can be leveraged to temporarily store thermal energy. However, the desired phase (mirabilite, hosting 10 water molecules) is hindered by the undesired formation of the metastable heptahydrate. In this work, we systematically map the nucleation and dissolution of both phases and discover that mirabilite nucleates very close to the formation temperature of ice, and cannot be stimulated via heterogeneous nucleation sites. This work highlights the need to gain control over the controlled nucleation of this salt to capitalise on its major potential for thermal energy storage.